|
Additionally, FDA took action this week to authorize boosters for 12-15 year olds – and I look forward to ACIP meeting on Wednesday to discuss this issue. First results from any randomized, controlled COVID-19 vaccine booster trial demonstrate a relative vaccine efficacy of 95. 
If you or your children are eligible for a third dose or a booster, please go out and get one as soon as you can. 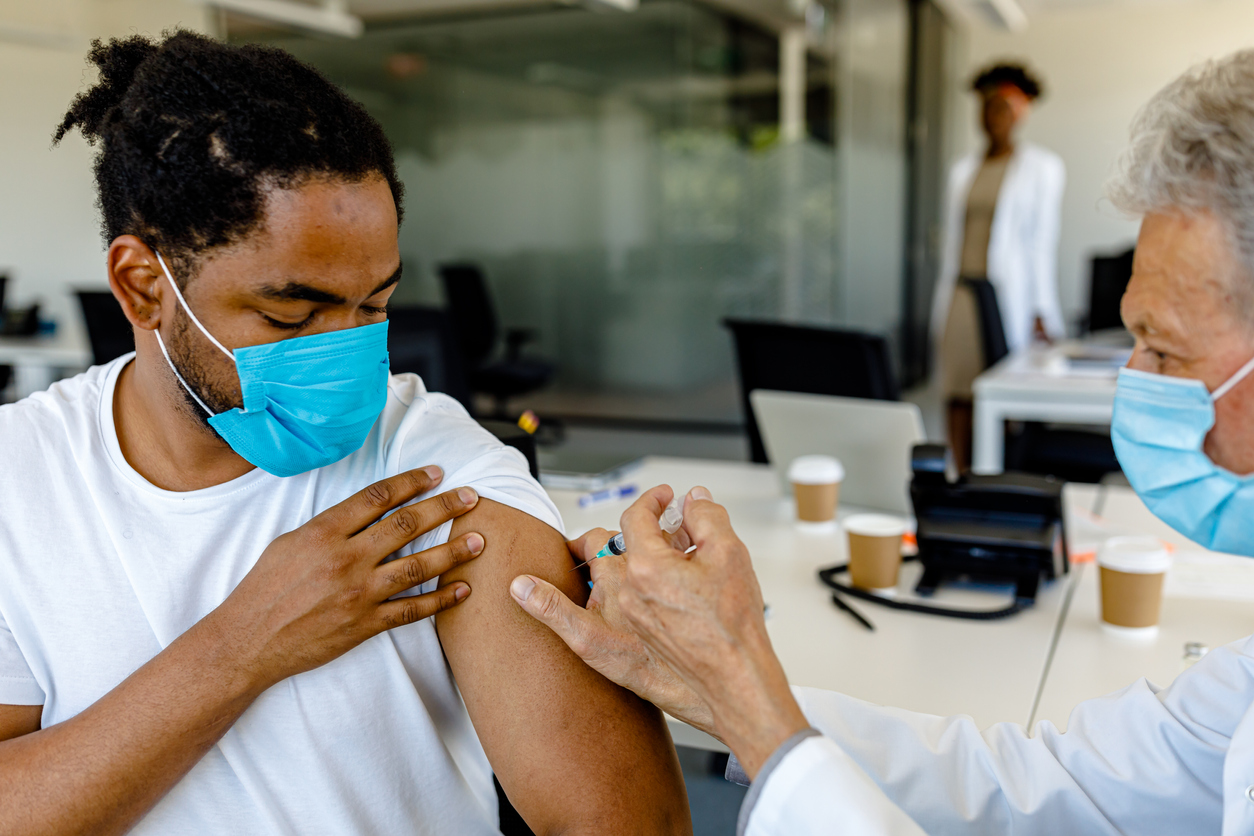
Following the FDA’s authorizations, today’s recommendations ensure people are able to get a boost of protection in the face of Omicron and increasing cases across the country, and ensure that the most vulnerable children can get an additional dose to optimize protection against COVID-19. Rochelle Walensky:Īs we have done throughout the pandemic, we will continue to update our recommendations to ensure the best possible protection for the American people. Other transient side effects would likely. I was planning to take a sick day just in case I. He received his first Pfizer doses in January and followed up with a Pfizer booster at a local pharmacy in late October. If 2 experienced severe fever, that would be 700,000 people. Luis Gallego, a 41-year-old early childhood education specialist, didn’t experience any noticeable side effects, with the exception of a sore arm, after any of his shots. But if the companies win regulatory approvals, they're aiming to supply vaccine to 35 million people globally by the end of December. The following is attributable to CDC Director, Dr. Fewer than 2 of recipients of the Pfizer and Moderna vaccines developed severe fevers of 39C to 40C. The safety of a single booster dose of the Pfizer-BioNTech COVID-19 Vaccine, Bivalent for individuals 12 years of age and older is based on safety data from a clinical study which evaluated a. /do0bihdskp9dy.cloudfront.net/07-17-2021/t_1f6d131ea4f248de92dd8dade933c7fa_name_file_1280x720_2000_v3_1_.jpg)
You cannot catch COVID-19 from the vaccine. The booster interval recommendation for people who received the J&J vaccine (2 months) or the Moderna vaccine (6 months), has not changed.Īdditionally, consistent with our prior recommendation for adults, CDC is recommending that moderately or severely immunocompromised 5–11-year-olds receive an additional primary dose of vaccine 28 days after their second shot. At this time, only the Pfizer-BioNTech COVID-19 vaccine is authorized and recommended for children aged 5-11. You can also take pain relief such as paracetamol to help you feel better. This means that people can now receive an mRNA booster shot 5 months after completing their Pfizer-BioNTech primary series. Today, CDC is updating our recommendation for when many people can receive a booster shot, shortening the interval from 6 months to 5 months for people who received the Pfizer-BioNTech COVID-19 Vaccine.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
